RITM SCENAR® technology vs micro-current and other E-stim devices comparison
RITMSCENAR technology is frequently mistaken for conventional Microcurrent Machine or TENS units (other E-stim)
Energetic treatments from SCENAR type devices have now been given to millions of people all over the world over the past 30 years without any recorded negative side effects.
You cannot harm someone with a RITMSCENAR® device treatment other than for the contraindications listed in the manual. The worst you can do is not be very effective.
RITMSCENAR® device is cleared by the FDA as a class 2 medical devices for delivering general noninvasive treatment via human skin areas for relief and management of chronic and acute pain, as adjunctive treatment in the management of post-surgical and post-traumatic pain.
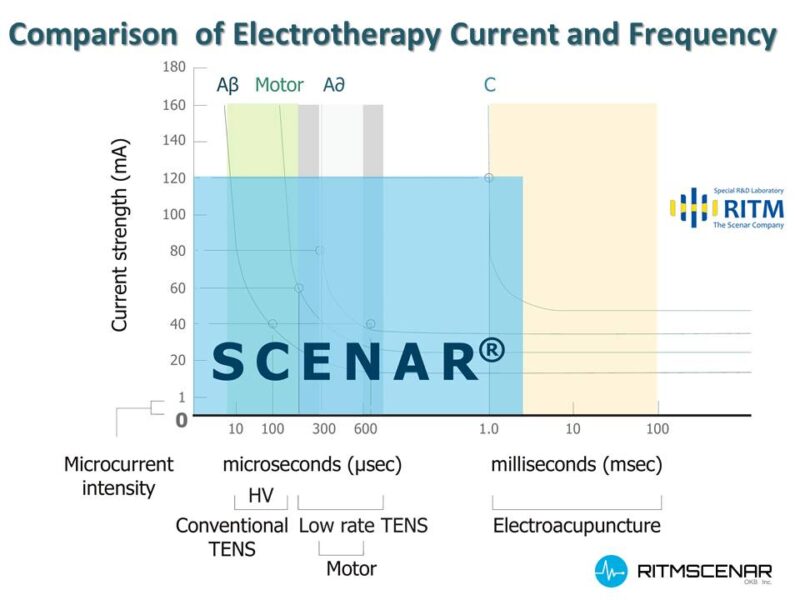
Simply said RITMSCENAR® is an advanced form of electro neurostimulation with “biofeedback” capability. It is NOT like a Micro current Machine or E-stim or TENS device in any way, shape, or form. The average Microcurrent Machine unit does not have the same unique output signature that newest RITMSCENAR® has.
RITM OKB ZAO engineers work hard on SCENAR® modernization to make our devices more effective and easier to use. First successful tests SCENAR were carried out back in 1986. This year RITM OKB ZAO celebrates 40th anniversary. SCENAR device is RITMSCENAR®, all others are copycats with no proof of SCENAR therapeutic effects.
RITMSCENAR® contains unique functions and Bio frequency sets to produce SCENAR impulses, which are transmitted by electrodes in the device through the skin to interface with the body`s internal peripheral nervous system for the purpose of therapeutic intervention.
RITMSCENAR®`s ability to communicate intelligently between the tissue and the device is what sets it apart from a conventional TENS unit, which is usually just feeding the same type of signal one-way into the tissue, without adjustments. When you do anything to the body consistently over time, it adapts, which is not what you want when attempting to treat pain. With the SCENAR®, not only will you experience longer-lasting (and more effective) pain relief immediately, but over time as well. During SCENAR treatments it is the neuropeptides that are the most important chemical modulators, neuropeptide producing nerve fibers make up more than 70% of the body`s neural tracts and can therefore be stimulated from many areas of the skin. The main goal of SCENAR® therapy is to induce the secretion of a sufficient amount of neuropeptides to relieve pain and initiate healing. The works evidenced the production of endorphins, enkephalins, and other neuropeptides in response to SCENAR therapy. The combination of these substances not only regulates the activity of the systems responsible for pain perception but also coordinates the functioning of many other body structures. It was also evidenced that in response to SCENAR therapy the sanogenesis mechanisms provide for the oxidative stress reduction, a decrease of the lipid peroxidation products level in the blood, restoring the activity of the antioxidant protection ferments, and decrease in the number and severity of inflammation processes. All medical devices are subject to stringent regulation by national and intergovernmental agencies. The approval process may include proof of efficacy, quality control, registration of manufacturers and distributors, labeling approval, and or other rigorous requirements.
FDA, CE mark, TGA, CANADA and MEXICO Health, South Korea, ISRAEL



1 Comment. Leave new
Please contact me. I would like to discuss by phone more specifics on the ifferences between scenar vs alpha Stim microcurrent.